|
Terms as meaning an increase in the number of microstates and therefore an increase in However, when we're using the equationĭeveloped by Boltzmann, we should think about these Using the word microstates, people will describeĪn increase in entropy as an increase in disorder or an increase in the dispersal Microstates decreases, that represents a decrease Of a system increases, that represents an increase in entropy, and if the number of According to this equation, entropy, symbolized by S, is equal to Boltzmann's constant, k, times the natural log of W, and W represents the number Now that we understand theĬoncept of microstates, let's look at an equationĭeveloped by Boltzmann that relates entropy to Is a number that's too high for us to even comprehend. So the number of microstatesĪvailable to this system of one mole of gas particles Moving from one microstate into another, into another, into another. The microscopic level, we see that the system is So from a macroscopic point of view, nothing seems to change. A good way to think aboutĪ microstate would be like taking a picture of Microscopic arrangement of positions and energies So going back to our boxes,īox 1, box 2 and box 3, each box shows a different To the kinetic energies of the particles. With an ideal gas here, by energies, we're referring Microscopic arrangement of all of the positions andĮnergies of the gas particles. Of each particle is equal to 1/2 mv squared, where m is the mass of each Particles are meant to represent the velocities of the particles. And the magnitude and theĭirection give a velocity. However, when we put anĪrrow on each particle, that also gives us the direction. 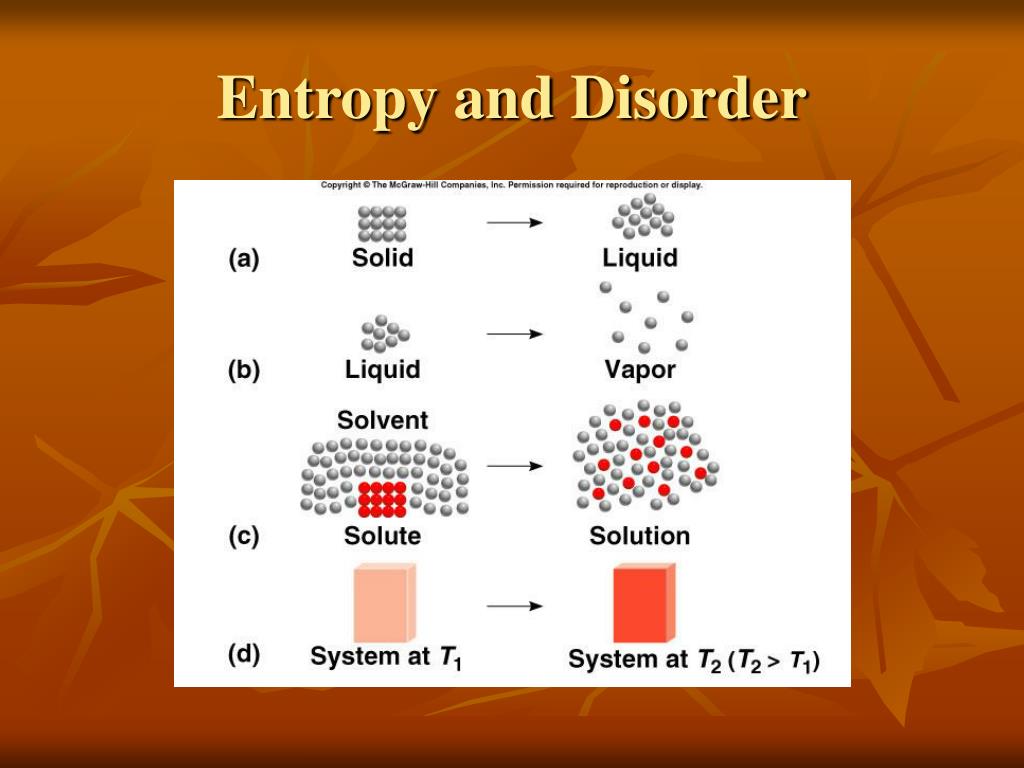
Of a particle tells us how fast the particle is traveling. Slightly different positions and the velocities might have changed. Particles in our system at one moment in time, in box 1, if we think about them atĪ different moment in time, in box 2, the particles might be in Slamming into each other and transferring energy from 
Slamming into the sides of the container and maybe Here in the first box, imagine these gas particles 
However, from a microscopic point of view, things are changing all of the time. So from a macroscopic point of view, nothing seems to be changing. Particles is at equilibrium, then the pressure, the volume, the number of moles, and the temperature all remain the same. Moles at a specific pressure, volume, and temperature. And to think about microstates, let's consider one mole of an ideal gas. The change of entropy of a system during a process is calculated using the equation: ΔS = S_final - S_initial, where ΔS is the change in entropy, S_final is the entropy of the system after the process is complete, and S_initial is the entropy of the system before the process started.Of entropy is related to the idea of microstates. However, a reaction that produces fewer molecules from more molecules will decrease the entropy of the system. For example, a reaction that produces more molecules from fewer molecules will typically increase the entropy of the system because there are more possibilities for arranging the molecules. The second law of thermodynamics states that the total entropy of a closed system always increases over time, which means that the system becomes more disordered or randomized over time.Īlso, chemical reactions can increase or decrease the entropy of a system. So, entropy is a measure of the number of possible arrangements of a system that are consistent with its overall properties, such as temperature, pressure, and volume. Entropy in simple terms is the disorder or randomness in a system.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
